
Many unproven stem cell-based therapies cost thousands of dollars to patients and are not covered by insurance. 4 Further, adverse events are likely underreported because these products are not FDA approved or regulated. The Pew Charitable Trusts gathered 360 reports of adverse events related to unapproved stem cell therapies, including 20 cases that caused death. A 2017 report described one Florida clinic that blinded patients with stem cell eye injections. For example, injected cells can multiply into inappropriate cell types or even dangerous tumors. The risks range from administration site reactions to dangerous adverse events. These unproven, unregulated stem cell treatments carry significant risk. However, these clinics almost always work without FDA regulatory approval and outside of legitimate clinical trial approaches. Clinics may provide misleading information and advertise their practice as running clinical trials. 
Some manufacturers offer stem cell-based derived products, such as “biologic eye drops” made with placenta extract or amniotic fluid to treat dry eye. 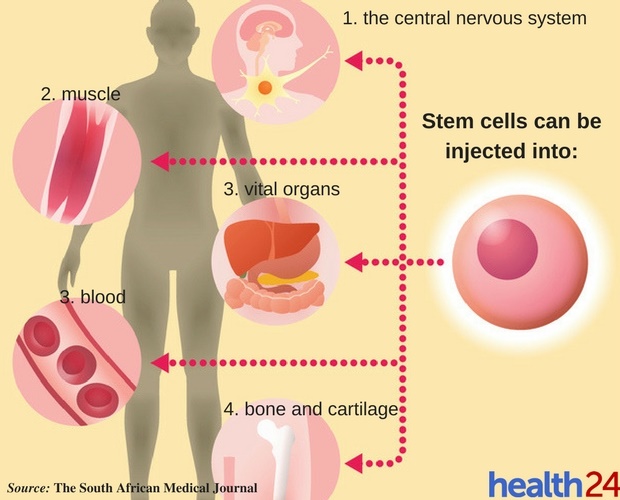
They may extract a patient’s own stem cells, concentrate or modify the cells, and then re-inject them. The clinics may mimic legitimate practices. However, at this time, rogue clinics, driven by profits, are taking advantage of patients desperate for cures and are claiming dramatic results, often exaggerated in sensational media testimonials. Over time, we expect that breakthroughs will continue with stem cell-based therapies for many conditions. 2 Stem cell-based therapies are also being explored for neurodegenerative diseases such as stroke and Alzheimer’s disease, and for countless other conditions. Since the early 2000s, stem cell-based therapies have been explored in many eye diseases, including age-related macular degeneration and glaucoma. 1 Other approved treatments include dental uses for gum and tissue growth and in skin for burns. These include bone marrow transplants, which have been transformational for many cancer patients, and therapies for blood and immune system disorders. Over the past three decades, the Food and Drug Administration (FDA) has approved several stem cell-based products. They offer a theoretically unlimited source of repair cells and/or tissues. These cells have the potential to develop into many different types of cells in the body. Stem cell-based therapies include any treatment that uses human stem cells. When communicating to the public about stem cell-based therapies, it is important to put any treatment claims in context. At the same time, thousands of rogue clinics have sprung up across the U.S and around the world, offering stem cell-based therapies before being tested for safety and efficacy. In recent years, the potential of stem cell-based therapies to treat a wide range of medical conditions has given hope to patients in search of novel treatments or cures. Wai, MD, Theodore Leng, MD, MS, and Jeffrey Goldberg, MD, PhD, Byers Eye Institute at Stanford, Stanford University School of Medicine, Palo Alto, CA
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
